DOCUMENT TITLE: |
Clinical Records Policy |
DOCUMENT NUMBER: |
ELHT/C013 Version 5.4 |
DOCUMENT REPLACES: |
Version 5.3 |
LEAD EXECUTIVE DIRECTOR DGM: |
Director of Finance, Information & Planning (SIRO) |
AUTHOR(S): |
Directorate Manager Centralised Outpatients & Administration Service |
TARGET AUDIENCE: |
All Trust Personnel |
DOCUMENT PURPOSE: |
The Clinical Records Policy sets out the standard required for clinical record keeping, and details the specific requirements relating to health records management within the organisation to ensure the delivery of an effective high quality clinical records service. This policy also sets out the current legislation relating to health records. |
To be read in conjunction with: |
|
SUPPORTING REFERENCES: |
|
Committee/Group |
Date |
|
Consultation |
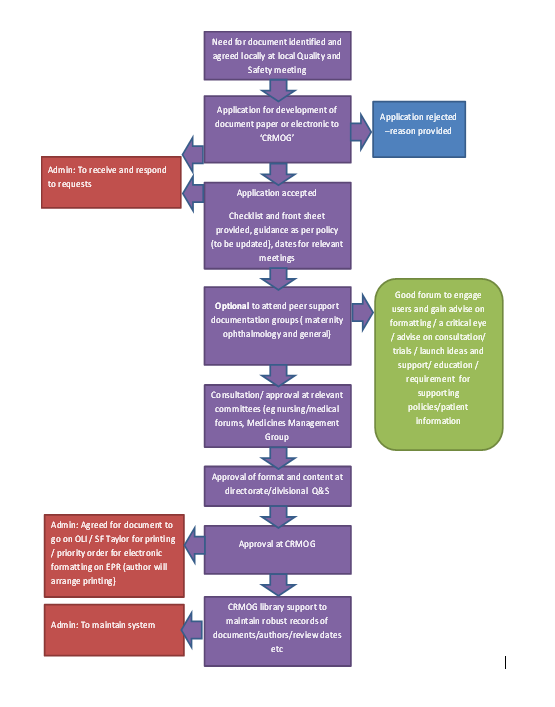
Clinical Records Modernisation and Oversight Group |
July 2021 |
Approval Committee |
Clinical Records Modernisation and Oversight Group |
July 2021 |
|
Document approved date |
August 2021 |
|
|
Next review date |
April 2024 |
|
|
Amendments |
V5 - Policy updated to reflect inclusion of community |
|
|
records following transfer of community services. |
||
|
V5.1 – Policy updated to reflect titles/meetings changes |
||
|
V5.2 – Policy updated to reflect the introduction of the |
||
|
Information Governance Alliance, Record Management Code of |
||
|
Practice for Health and Social Care 2016 following the |
||
|
withdrawal of the DOH Records Management Code of Practice |
||
|
Parts 1 & 1. |
||
|
Title changes |
||
|
Inclusion of the HRSG Terms of Reference V5.3 – Policy updated to reflect the following changes: - guidance in relation to the destruction of patient records in light of the Independent Enquiry into Child Sexual Abuse (IICSA) - TOR - Clinical Audit Schedule Introduction of General Data Protection Act (GDPR) in relation to Subject Access Requests V5.4 – Policy updated to reflect the following - Removal of ‘TOR’ and ‘Audit Schedule’ - Renaming of the HRSG to Clinical Records Modernisation and Oversight Group - Title changes - Revision to supporting references - Amendments to section 3 ‘Legal Obligations that apply to records’ - Amendment to section 14’ Records Security and storage – inclusion of off-site record storage’ - Inclusion of section 12 ‘National Data Opt Out’ - Inclusion of section 13 ‘Guidance in relation to the Gender Recognitions Act 2004 – records management’ - Inclusion of section 16 ‘Records access and auditing’ - Revision to Appendix one –‘Updated records management code of practice 2020’ |
Health Records Retention Schedule – Please refer to the Records Management Code of Practice 2020 pages 51 to 71 as per the link below.
Records Management Code of Practice - NHSX
See Section 14.3. Regarding the temporary suspension of the destruction of health records
Principles to be used in Determining Policy Regarding the Retention and Storage of Essential Maternity Records
British Paediatric Association
Royal College of Midwives
Royal College of Obstetricians and Gynaecologists
United Kingdom Central Council for Nursing, Midwifery and Health Visiting
Joint Position on the Retention of Maternity Records
1. All essential maternity records should be retained. ’Essential’ maternity records mean those records relating to the care of a mother and baby during pregnancy, labour and the puerperium.
2. Records that should be retained are those which will, or may, be necessary for further professional use. ’Professional use’ means necessary to the care to be given to the woman during her reproductive life, and/or her baby, or necessary for any investigation that may ensue under the Congenital Disabilities (Civil Liabilities) Act 1976, or any other litigation related to the care of the woman and/or her baby.
3. Local level decision making with administrators on behalf of the health authority must include proper professional representation when agreeing policy about essential maternity records.
’Proper professional’ in this context should mean a senior medical practitioner(s) concerned in the direct clinical provision of maternity and neonatal services and a senior practising midwife.
4. Local policy should clearly specify particular records to be retained AND include detail regarding transfer of records, and needs for the final collation of the records for storage. For example, the necessity for inclusion of community midwifery records.
5. Policy should also determine details of the mechanisms for return and collation for storage, of those records which are held by mothers themselves, during pregnancy and the puerperium.
6. List of maternity records retained should include the following:
6.1 Documents recording booking data and pre-pregnancy records where appropriate.
6.2 Documentation recording subsequent antenatal visits and examinations.
6.3 Antenatal in-patient records.
6.4 Clinical test results including ultrasonic scans, alpha-feto protein and chorionic villus sampling.
6.5 Blood test reports.
6.6 All intrapartum records to include, initial assessment, partograph and associated records including cardiotocograph.
6.7 Drug prescription and administration records.
6.8 Postnatal records including documents relating to the care of mother and baby, in both the hospital and community setting.
|
Aspect of compliance being measured or monitored. |
Individual responsible for the monitoring |
Tool and method of monitoring |
Frequency of monitoring |
Responsible Group or Committee for monitoring |
|
Outpatient Casenote Availability and Trends |
Deputy Health Records Manager |
Information is extracted from PAS via business intelligence tool |
Monthly |
Clinical Records Modernisation and Oversight Group (CRMOG) |
|
Datix Records related incidents ‘Problems with Patient Records’ |
Health Records Manager |
Datix Reporting tool – Action Plan |
Monthly |
Clinical Records Modernisation and Oversight Group (CRMOG) |
|
Recent Care Audit /Basic record keeping standards |
Clinical Audit with Medical Staff |
Review of case notes |
Annual |
Clinical Records Modernisation and Oversight Group (CRMOG) |
|
Case Note Storage |
Health Records Manager |
Audit tool / SOP |
(previously Annual) to move to Monthly |
Clinical Records Modernisation and Oversight Group (CRMOG) |
|
Rationale for Clinical decision making, |
|
To be agreed |
To be agreed |
|
|
Correct consultant (new 2021) |
Ward managers/ Consultants |
On ward EPTS/ Case notes |
Monthly |
Individual ward action plan |
|
NAPF documentation audit (new 2021) |
NAPF team |
Trust wide documentation audit |
One off |
Individual ward action plan |
|
IG audit |
IG team |
Area visit and observations |
|
Individual ward action plan |